Kingpin Carbon – Why?

By Rhiannonw
- 497 reads
Beautiful, glittering, hard, hard diamond held aloft
or messy, useful, charcoal soft
– differing internal structure
as if each atom has four 'arms' that reach
and clasp more or its own,
hooking up in tight crystals, or thin fragile plates,
or catch and hold other kinds – in strings or rings,
as building blocks,
or energy-storers,
or fascinating, complicated, large twisted exact shapes
that are like 'keys' or 'locks'
to fit together to trigger specific reactions,
interactions.
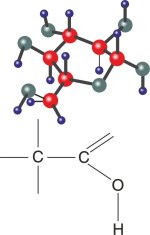
The atom that makes possible the complex chemistry of life:
[IP: Choose a chemical element]
- Log in to post comments
Comments
Fantastic
Fantastic. The atomic structure - both are of pure carbon element, but graphite a planar carbon (2D-lattice) and diamond (a 3D structure). One would obviously not try it Rhiannon, but I've wondered could a diamond actually burn in oxygen like graphite?
The one being of the very softest on the Mohr scale, while the other is the hardest known substance.
Silicon also makes this kind of very ordinary SiO3 of 3D lattices as rubies, emerald, saphyre etc with different impurities.. Quarts crystal too, I don't know how it works.
Your explanations are excellent, sounds like a born teacher (mother, grandmother! Must be)
Goodnight sleep well! Tom
- Log in to post comments